 
There are exceptions however, such as water's density increasing between 0☌ and 4☌.īelow is a table of units in which density is commonly expressed, as well as the densities of some common materials. Iron has three major isotopes: 54Fe (atomic mass 53.9396 u abundance 5.85), 56Fe (atomic mass 55.9349 u abundance 91.75), and 57Fe (atomic mass. Increases in temperature tend to decrease density since the volume will generally increase. Question: em 20 There are four naturally occurring isotopes of iron: Fe, 56 Fe, Fe and Fe Part A How many protons, neutrons, and electrons are in Fe Express your answers as integers separated by commas. An increase in pressure decreases volume, and always increases density. The elements of the periodic table sorted by atomic mass 98, Technetium 101.07, Ruthenium 102.9055, Rhodium 106.42, Palladium 107.8682, Silver. The atomic weight given for an element on the Periodic Table of Elements is actually its average atomic mass. However, when regarding gases, density is largely affected by temperature and pressure. The average atomic mass of iron is 55.85. In the case of solids and liquids, the change in density is typically low. Note that density is also affected by pressure and temperature. 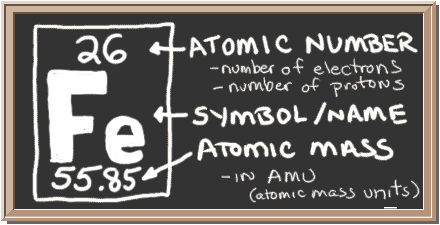
The atomic mass of natural elements represents an average mass of the atoms. To calculate the average atomic mass, multiply the fraction by the mass number for each isotope, then add them together. It is useful to carefully write out whatever values are being worked with, including units, and perform dimensional analysis to ensure that the final result has units of mass For example, an oxygen atom has 8 protons and an iron atom has 26 protons. The chlorine isotope with 18 neutrons has an abundance of 0.7577 and a mass number of 35 amu. There are many different ways to express density, and not using or converting into the proper units will result in an incorrect value. However, it is important to pay special attention to the units used for density calculations. The calculation of density is quite straightforward.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
